Open the Atomic Weight Calculator to explore the relationship between the isotopic abundances of an element and the element's atomic weight. For now, use the Atomic Weight Calculator to find the atomic weight of carbon in a sample. Click on the large element symbol to pull up the Periodic Table and select carbon. The isotopic abundances are, by default, set to the approximate abundances that are found in nature. What is the atomic weight of carbon?
Now, use the formula below to determine the atomic weight of carbon in a particular sample without using the learning tool, and then check your answer.
Recall the information that is required to calculate an atomic weight. First, you need to know the number of stable isotopes of carbon. Carbon has two stable isotopes: carbon-12 and carbon-13.
You also need to know the relative abundance of each of the isotopes. Assume that in the sample, the abundance of carbon-12 is 0.9893 and the abundance of carbon-13 is 0.0107. These abundances are very similar to those that can be found in nature.
To calculate the atomic weight of carbon, you must also know the relative atomic masses of each of carbon's stable isotopes. The relative atomic mass of carbon-12 is defined as exactly 12 and the relative atomic mass of carbon-13 is 13.00335. Now, you can calculate the atomic weight of carbon in the sample.
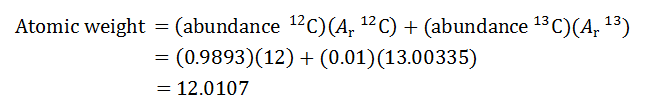
For this sample, the atomic weight of carbon is 12.0107. Does this match the atomic weight of carbon given by the learning tool?