While all isotopic abundances have some variation, you should notice that not all the elements have listed intervals for their atomic weights. Currently, IUPAC has separated the elements in the periodic table into four categories.
|
The pink category displays elements whose atomic weight is an interval. These intervals are not uncertainties due to imprecise measuring devices, but represent the natural variation inherent in samples of these elements. |
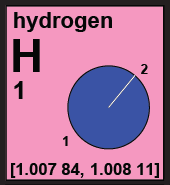 |
|
The yellow category identifies elements with two or more isotopes that determine atomic weight, but where the atomic weight interval has not been determined by IUPAC, or the variation in isotopic abundances is too small to affect the atomic weight significantly, and so the atomic weight is listed as a single value with an uncertainty within parentheses. |
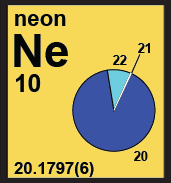 |
|
The blue category identifies elements with only isotope that determines atomic weight. The atomic weight of these elements are constants of nature, and the value is given with an uncertainty within parentheses. |
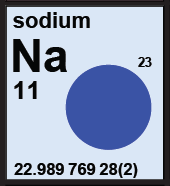 |
|
The white category identifies elements with no standard atomic weight because all of the isotopes are radioactive in normal materials, and no atomic weight for particular samples can be determined. |
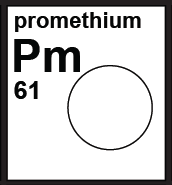 |
Open the IUPAC Periodic Table of the Elements and Isotopes Learning Tool. Select elements and explore the 'more information' tab to find an isotope in each category that is vitally important to society.
How many elements are given atomic weight intervals?